Now Reading: Scientists Reveal How FTL1 Is Linked with Aging Brain
-
01
Scientists Reveal How FTL1 Is Linked with Aging Brain
Scientists Reveal How FTL1 Is Linked with Aging Brain
The aging brain is increasingly linked to a protein called FTL1, which plays a critical role in iron metabolism and cognitive decline. Scientists have found that higher levels of FTL1 are associated with memory loss, weakened brain connections, and slower brain function, while reducing this protein may help reverse some aging effects.
What Is FTL1 and Why It Matters in the Aging Brain
FTL1, also known as ferritin light chain 1, is a protein that plays a central role in how the brain stores and manages iron. Iron is essential for normal brain function, as it supports processes like energy production and neurotransmission.
However, maintaining the right balance is crucial, since too much or improperly regulated iron can lead to cellular damage. FTL1 is a component of ferritin, the primary iron storage system in cells, and it helps regulate iron detoxification and electron transfer within brain tissue.
In the context of the aging brain, FTL1 has gained attention because of its direct connection to cognitive health. Rather than being linked to data transmission or technology, as sometimes misunderstood, FTL1 is purely a biological protein that influences how brain cells function over time. Its importance lies in how it affects iron balance, which in turn impacts neuronal health, memory, and overall brain performance.
The Discovery of FTL1 as a Driver of Brain Aging
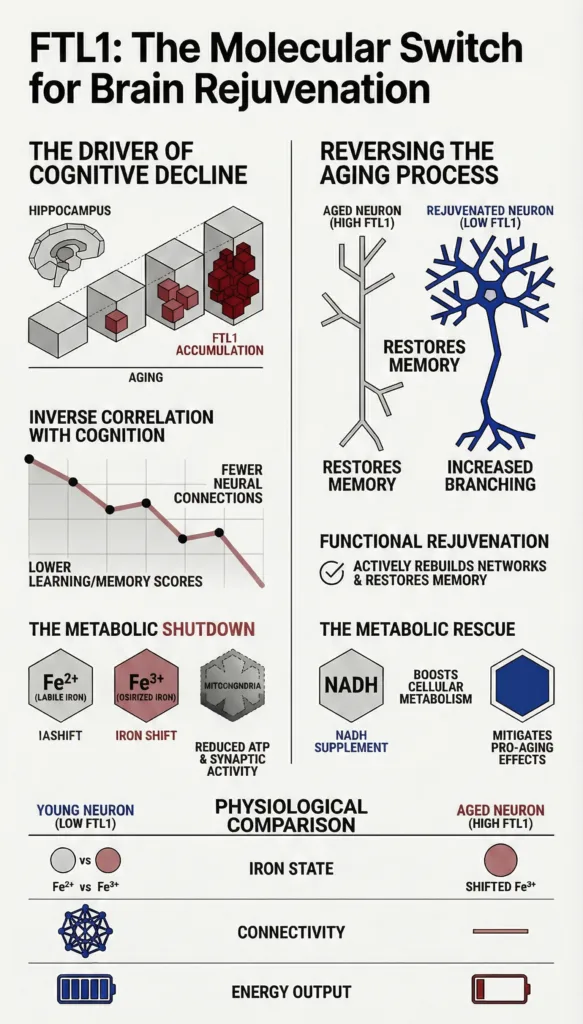
Recent scientific research has highlighted FTL1 as a major factor in brain aging, particularly in the hippocampus, which is the region responsible for learning and memory. Scientists studying aging in mice observed that FTL1 levels consistently increased as the animals grew older. Among many proteins analyzed, FTL1 stood out as the only one that showed a strong and consistent rise with age.
This increase was not just a random occurrence. It closely correlated with measurable declines in cognitive performance. Older mice with higher levels of FTL1 performed worse on memory tasks, indicating a direct link between the protein and brain function. This discovery positioned FTL1 as a key molecular driver of aging in the brain, rather than just a byproduct of the aging process.
FTL1: The Iron-Associated Driver of Brain Aging and Degeneration
The identification of FTL1 as a central factor represents a significant step forward in understanding how aging affects the brain at a molecular level. It provides a clear target for further research and potential therapeutic development.
How FTL1 Affects Brain Function and Cognitive Health
FTL1 influences the aging brain through several interconnected mechanisms that ultimately impair cognitive function. One of the most significant effects is the weakening of connections between neurons. These connections, known as synapses, are essential for communication within the brain. When FTL1 levels rise, these connections become less effective, leading to reduced information processing and memory retention.
In addition to weakening neural connections, FTL1 contributes to memory decline. Studies have shown that animals with elevated levels of this protein experience noticeable reductions in their ability to learn and recall information. This decline is not subtle, as it affects performance in cognitive tests that measure memory and problem solving abilities.
Another critical impact of FTL1 is its influence on the structure of brain cells. When researchers artificially increased FTL1 levels in younger subjects, their brain cells began to resemble those of older individuals. Instead of forming complex branching networks, neurons developed simpler structures with fewer connections. This structural simplification limits the brain’s capacity to adapt and process new information.
FTL1 also disrupts how brain cells produce and use energy. Proper cellular metabolism is essential for maintaining brain activity, and any slowdown can have serious consequences. Elevated FTL1 levels interfere with metabolic processes in the hippocampus, reducing the efficiency of energy production. This metabolic disruption further contributes to cognitive decline and overall brain aging.
The Role of Iron Metabolism in Brain Aging
Iron metabolism plays a crucial role in maintaining brain health, and FTL1 is deeply involved in this process. The brain requires iron for various functions, including oxygen transport and the synthesis of neurotransmitters. However, iron must be carefully regulated to prevent harmful effects.
FTL1 contributes to the management of iron by participating in its storage and detoxification. As part of the ferritin complex, it helps convert iron into forms that are less reactive and safer for cells. However, when FTL1 levels become too high, this balance is disrupted. The protein alters iron oxidation states, which can lead to increased oxidative stress and cellular damage.
This imbalance affects neurons directly, impairing their ability to function properly. Over time, the accumulation of these effects contributes to the aging of the brain. The relationship between FTL1 and iron metabolism highlights the importance of maintaining proper biochemical balance for long term cognitive health.
Structural and Functional Changes in the Aging Brain
As FTL1 levels increase, both the structure and function of the brain undergo noticeable changes. Neurons lose their complex branching patterns, which are essential for forming strong and diverse connections. Instead, they develop shorter and simpler extensions, reducing their ability to communicate effectively.
Functionally, these structural changes translate into weaker neural networks. The brain becomes less efficient at processing information, leading to slower thinking and reduced memory capacity. Tasks that once required minimal effort become more challenging, reflecting the overall decline in cognitive performance.

These changes are not limited to natural aging. Experimental studies have shown that artificially increasing FTL1 levels in younger subjects can accelerate these structural and functional alterations. This finding reinforces the idea that FTL1 is not just associated with aging but actively drives the process.
Can Reducing FTL1 Reverse Brain Aging
One of the most promising aspects of FTL1 research is the potential for reversing the effects of brain aging. Scientists have found that lowering FTL1 levels in older subjects leads to significant improvements in brain function. These improvements are not merely preventive but appear to restore lost capabilities.
In experimental settings, reducing FTL1 resulted in stronger connections between neurons and better performance in memory related tasks. The changes were described as a true reversal of impairments, rather than just slowing down the aging process. This suggests that the aging brain may retain a degree of plasticity that can be reactivated under the right conditions.
The possibility of reversing cognitive decline opens new avenues for research and treatment. It challenges the traditional view that aging related brain changes are irreversible and provides hope for developing interventions that restore cognitive function.
Metabolism, Energy, and the FTL1 Connection
The link between FTL1 and cellular metabolism is another critical area of study. Brain cells require a constant supply of energy to function properly, and any disruption can have widespread effects. Elevated FTL1 levels have been shown to slow down metabolic processes in the hippocampus, reducing the efficiency of energy production.
This slowdown affects the brain’s ability to maintain normal activity levels. Neurons become less responsive, and their ability to form and maintain connections is compromised. As a result, cognitive functions such as memory and learning are negatively impacted.
Researchers have explored ways to counteract these effects by boosting cellular metabolism. Treatments that enhance metabolic activity have been shown to reduce the negative impact of FTL1. For example, compounds that support energy production can prevent the cognitive decline associated with high FTL1 levels. This approach highlights the importance of metabolic health in maintaining brain function.
Therapeutic Potential and Future Research Directions
The discovery of FTL1 as a key driver of brain aging has significant implications for the development of new therapies. By targeting this protein, scientists may be able to create treatments that not only slow down cognitive decline but also reverse it.
One promising strategy involves reducing FTL1 levels directly. This approach has already shown success in experimental models, where it led to improved memory and stronger neural connections. Another approach focuses on supporting cellular metabolism to counteract the effects of FTL1. Enhancing energy production in brain cells may help maintain cognitive function even in the presence of elevated protein levels.
These findings suggest that FTL1 could become a central target in the fight against age related cognitive decline. However, further research is needed to understand its role in humans and to develop safe and effective treatments. The transition from laboratory studies to clinical applications will require careful investigation and validation.
Conclusion
The aging brain is a complex system influenced by multiple factors, but the discovery of FTL1 has provided a clearer understanding of how cognitive decline occurs at a molecular level. This protein plays a critical role in iron metabolism, neuronal structure, and cellular energy production, all of which are essential for maintaining brain health. The ability to reverse some of its effects offers new hope for addressing age related cognitive decline. As research continues, FTL1 may become a key focus in developing therapies that improve brain function and quality of life in aging populations.
Frequently Asked Questions
What is FTL1 in the aging brain
FTL1 is a protein involved in iron storage and metabolism in the brain. It plays a significant role in cognitive decline as its levels increase with age.
How does FTL1 affect memory
Higher levels of FTL1 weaken connections between neurons and disrupt brain function, leading to reduced memory and learning ability.
Is FTL1 related to technology or data transmission
No, FTL1 is a biological protein and not related to any data transmission or networking technology.
Can reducing FTL1 improve brain function
Yes, studies in experimental models show that lowering FTL1 levels can restore neural connections and improve memory performance.
Why is iron metabolism important in brain aging
Iron supports essential brain functions, but imbalance can cause damage. FTL1 regulates iron, and its disruption contributes to aging effects.
Are there treatments targeting FTL1
Research is ongoing, and early findings suggest that targeting FTL1 or improving cellular metabolism may lead to future therapies for cognitive decline.
Also read: 6 Secrets to Learn Faster, Backed by Neuroscience

Dony Garvasis is the founder of Search Ethics, a freelance SEO consultant, and a passionate technology blogger. With over 8 years of experience in SEO, blogging and content marketing, I provide expert content on Tech, digital marketing, SEO, Artificial intelligence, gadgets, science, automobiles, lifestyle, tips, tutorials and much more. My mission is simple: Ethical Search, Genuine Results! I will make sure people everywhere get trustworthy and helpful information.